Boltzmann's equation describes the evolution of molecules in a rare gas.
If no external forces are present ( ![]() ) then,
after a long time, the gas should reach an equilibrium state. This can
be seen [14, 11] by considering the function
) then,
after a long time, the gas should reach an equilibrium state. This can
be seen [14, 11] by considering the function
Differentiating equation (2.19) with respect to time we get
![]()
Substituting ![]() from equation (2.3), with
from equation (2.3), with
![]() ,
and using
equation (2.16) we get
,
and using
equation (2.16) we get
Now, following [14],
consider the first term on the right hand side of
equation (2.21). Since ![]() and
and ![]() are
independent variables we can write
are
independent variables we can write ![]() as
as
![]() and using Gauss's
theorem we can write
and using Gauss's
theorem we can write
![]()
where ![]() is the surface enclosing the volume
is the surface enclosing the volume
![]() . Now
. Now
![]() so the
first term on the right-hand side
of equation (2.21) vanishes. This leaves
so the
first term on the right-hand side
of equation (2.21) vanishes. This leaves
Now consider the reverse of the collision: particles with velocity
![]() and
and ![]() colliding
and moving off with velocities
colliding
and moving off with velocities ![]() and
and
![]() . For this collision we have
. For this collision we have
since ![]() .
Summing equations (2.23) and (2.24) and dividing by two gives
.
Summing equations (2.23) and (2.24) and dividing by two gives
Changing the dummy variables ![]() and
and ![]() we get
we get
Finally summing equations (2.25) and (2.26) and dividing by two gives
Now ![]() and all the other
terms in the integrand of equation (2.27) are positive so
and all the other
terms in the integrand of equation (2.27) are positive so
This means that H can never increase and is known as Boltzmann's
H-theorem. It can also be shown that H is bounded below [11].
We know that ![]() converges because
the total energy of the molecules must converge. Thus either H converges or
converges because
the total energy of the molecules must converge. Thus either H converges or
![]() more rapidly than
more rapidly than ![]() as
as
![]() . In the later case
. In the later case
![]() more rapidly than
more rapidly than ![]() which implies
that H converges. Since H can never increase but tends to a finite
limit the finite limit must correspond to
which implies
that H converges. Since H can never increase but tends to a finite
limit the finite limit must correspond to ![]() . This is
only possible, see equation (2.27), if
. This is
only possible, see equation (2.27), if
![]()
This condition is known as detailed balance [11] and can be expressed equivalently as
![]()
Thus if ![]() is the equilibrium distribution then
is the equilibrium distribution then
![]() is a collision invariant and so must be of the form
is a collision invariant and so must be of the form
![]()
where ![]() are the collision invariants defined below
equation (2.7) and
are the collision invariants defined below
equation (2.7) and ![]() are constants. This can be
re-written [11]
are constants. This can be
re-written [11]
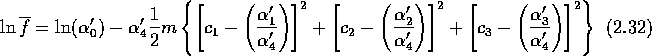
where ![]() and
and ![]() . With
. With
![]() where
where ![]() we can write
we can write
This is the Maxwell distribution function for a gas [11, 12, 14] and it describes the equilibrium state of the distribution function f. The form of the constants can be found by substituting equation (2.33) into equation (2.5) to give the more common form of the Maxwell distribution function [11, 12]
The H-theorem states that the distribution function f must tend
towards its equilibrium state ![]() .
The entropy S(t) of the system (which
is a non-decreasing function of time) is given by [11, 14]
.
The entropy S(t) of the system (which
is a non-decreasing function of time) is given by [11, 14]
![]()