Rather than considering the densities of the two fluids, ![]() and
and ![]() ,
the total density,
,
the total density, ![]() , and the density difference or
order parameter,
, and the density difference or
order parameter,
![]() , are considered.
Two distribution functions
, are considered.
Two distribution functions
![]() and
and ![]() are then used to describe the population of
are then used to describe the population of
![]() and
and ![]() respectively
on each of the i links. The evolution
of both distribution functions are governed by the single relaxation time
Boltzmann equations:
respectively
on each of the i links. The evolution
of both distribution functions are governed by the single relaxation time
Boltzmann equations:
and
The equilibrium distributions ![]() and
and
![]() take the general
form
take the general
form
and
The coefficients ![]() and
and
![]() are chosen to satisfy the following equations.
The equilibrium distribution must satisfy the conservation of mass and
momentum equations:
are chosen to satisfy the following equations.
The equilibrium distribution must satisfy the conservation of mass and
momentum equations:
while the higher moments of ![]() and
and ![]() are
defined
are
defined
so that the resulting continuum equations have the correct form for a
binary fluid [9, 64].
Here ![]() is the mobility and
is the mobility and ![]() is the chemical-potential difference between the two components.
The pressure tensor
is the chemical-potential difference between the two components.
The pressure tensor ![]() and the chemical potential
and the chemical potential
![]() determine the thermodynamical properties of the model and
are determined by the free energy [9, 38, 14, 65].
Orlandini et al.
[9] choose the free energy describing the
simplest possible binary fluid; two ideal gases with a repulsive
interaction energy. This corresponds to a free energy [9, 38]
determine the thermodynamical properties of the model and
are determined by the free energy [9, 38, 14, 65].
Orlandini et al.
[9] choose the free energy describing the
simplest possible binary fluid; two ideal gases with a repulsive
interaction energy. This corresponds to a free energy [9, 38]
![]()
where T is the temperature,
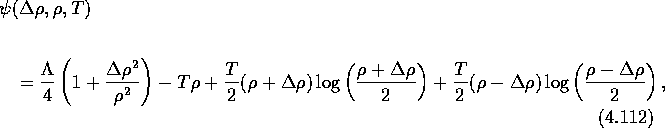
![]() is the interfacial energy
and
is the interfacial energy
and ![]() measures the strength of the interaction. For
measures the strength of the interaction. For ![]() the mixture separates into two phases.
With this definition of the free energy the chemical potential and the
pressure tensor are given by [9, 38, 14, 65]
the mixture separates into two phases.
With this definition of the free energy the chemical potential and the
pressure tensor are given by [9, 38, 14, 65]
![]()
and
were